[Major Contents]
Welcome English / 日本語
People/メンバーの紹介
Research subjects/研究の概要
Seminar/研究室ゼミ
Prospective Students/学生募集
Fee for Analysis/依頼分析
スポンサー/Sponsorship
[Others]
安定同位体比を用いた研究の基礎知識
海水魚とサンゴの飼育: 概要 と 注意点
水槽の説明と自作 と ギャラリー
コラム:研究者を志す皆さんへ
研究室の写真: Instagram と 写真ギャラリー
Welcome
We would love to go steady with basic and applied studies one at a time, for better understanding the 'complex' biogeochemical processes in biosphere. Currently, we are particularly focusing to the following three challenges:
- Illustration of 'energetic flux' in a single organism, an interaction between organisms (e.g., symbiosis, parasitism, etc.), and a trophic transfer along food webs;
- Illumination of ‘key process’ responsible for the change in the stable isotope ratios (D/H, 13C/12C, 15N/14N, etc.) of organic compounds with respect to organisms’ physiology; and
- Evaluation of ‘universality' based on accurate understanding of 'generality vs. predictable fluctuation' or 'homogeneity vs. heterogeneity’ in the change in the isotope ratios of organic compounds in ecological and geochemical samples.
To do that, we also enjoy the challenges for developing new analytical methods (and new methodologies) of compound- and position-specific isotope analyses of organic compounds in natural samples.
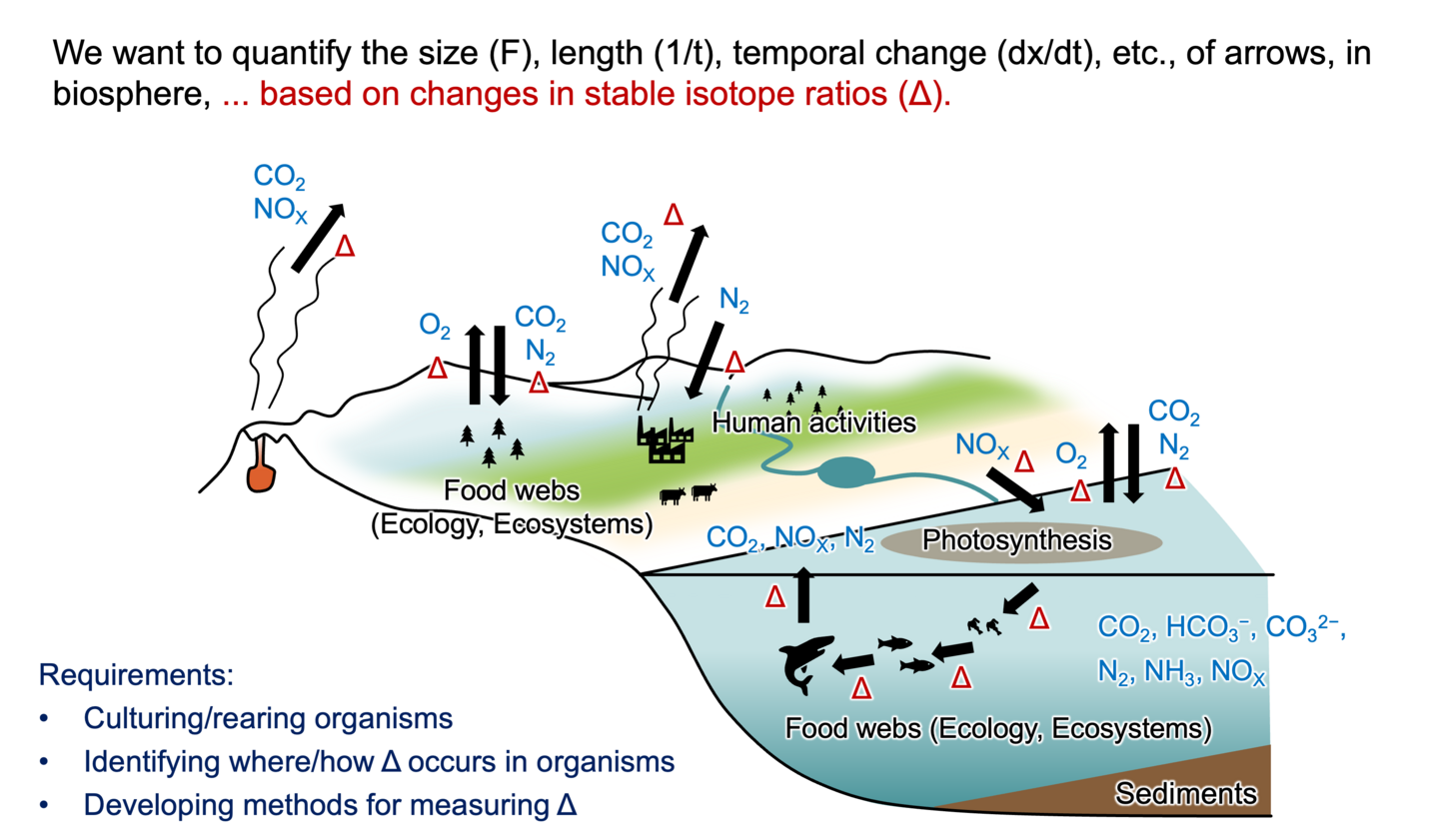
Isotope analysis of organic compounds
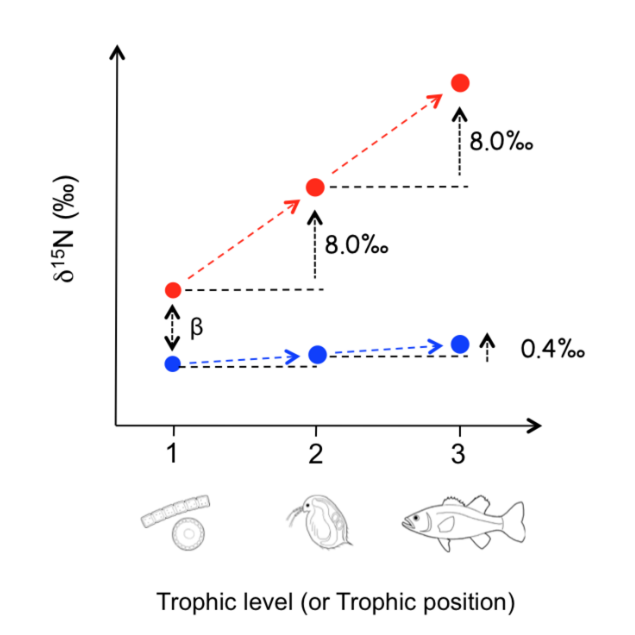
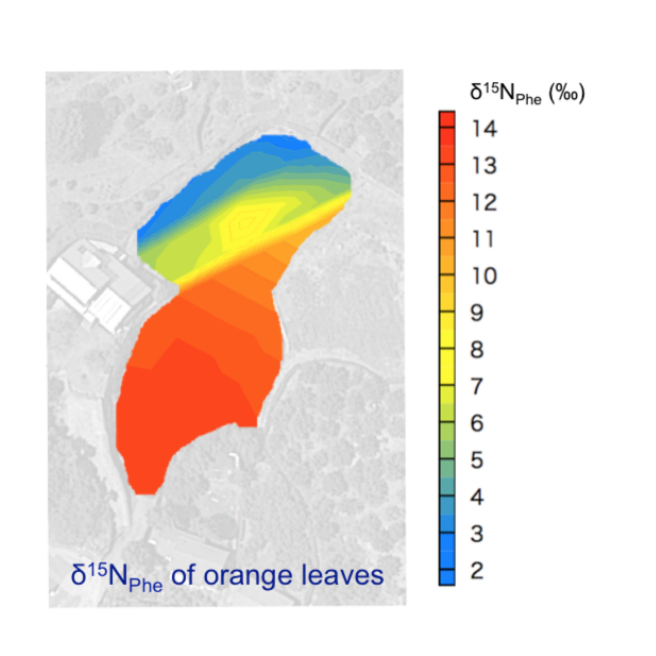
During the past three decade, the analysis of stable isotope ratios of individual organic compounds has been widely used in the study of biogeochemistry. While DNA analysis can visualize the biosphere in terms of species through genetic profiles, isotope analysis can visualize the biosphere in terms of the quantitative flux in physical and/or biochemical processes. The change in the isotope ratios (i.e., isotopic discrimination/fractionation) of organic compounds are frequently quantified by the kinetic isotope effect and flux of the processes, and the wide array of biochemical reactions within organisms creates diverse and distinctive isotope ratios among organic compounds. The isotope ratios of organic compounds therefore can be useful for characterizing and quantifying any given processes in biosphere.
Key process responsible for the change in the stable isotope ratios
The process driving isotopic discrimination/fractionation during biosynthetic and breakdown pathways in organisms is defined as 'key process'. An identification of 'key process' is therefore indispensable for a fundamental understanding of 'universality' of the isotope ratios found in organisms, as well as for validating the accuracy and precision of the isotope evidence in application studies.
People
Research Interests







- Welcome (Engish)
- Welcome (日本語)
- People/メンバー紹介
- 研究室の方針・ゼミ
- Prospective Student/学生募集
- Fee for Analysis/分析依頼
- 記事
- 水槽
[Major Contents]
Welcome English / 日本語
People/メンバーの紹介
Research subjects/研究の概要
Seminar/研究室ゼミ
Prospective Students/学生募集
Fee for Analysis/依頼分析
スポンサー/Sponsorship
[Others]
安定同位体比を用いた研究の基礎知識
海水魚とサンゴの飼育: 概要 と 注意点
水槽の説明と自作 と ギャラリー
コラム:研究者を志す皆さんへ
研究室の写真: Instagram と 写真ギャラリー